
The Ultimate Guide to CBD Edibles: Benefits, Dosage & Best Gummies
Introduction to CBD Edibles CBD edibles are food products infused with cannabidiol (CBD), a non-intoxicating compound found in hemp plants.

Introduction to CBD Edibles CBD edibles are food products infused with cannabidiol (CBD), a non-intoxicating compound found in hemp plants.

Cannabidiol effects mostly fall into two categories: the immediate and the long term. Either of these can be good or

There was excitement as the Charlotte’s Web bill passed through the Florida senate in April. This excitement was followed with
Like the common misperception of hemp vs. marijuana, there has always been confusion between marijuana and hashish. While some people

If you are worried about your job due to hemp oil use, you are not alone in this situation. Thousands

Hemp oil for inflammation is an option that many people are trying to support conventional treatments. In addition to anecdotal
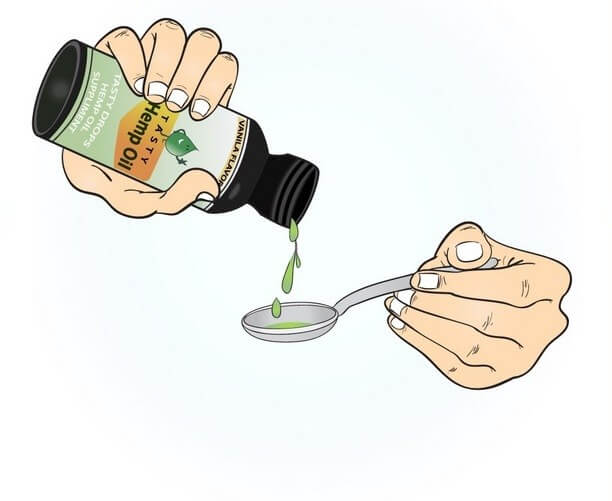
Before discussing cannabidiol (CBD) dosage, it makes sense to start with some basic cannabis and CBD facts. CBD is one of

The influence of the many CNN marijuana documentaries and articles cannot be underestimated. For starters, CNN is one of the

Hemp oil is derived from the seeds of the hemp plant. Hemp is a hardy and fast-growing plant regarded by
Marijuana is becoming increasingly popular as one of the arthritis herbal remedies used by hundreds of thousands of the over




FDA Disclaimer: These statements have not been evaluated by the Food and Drug Administration. Products sold by Healthy Hemp Oil are not intended to diagnose, treat, cure, or prevent any disease. The information on our website is intended to provide general information regarding our products and is not to be construed as medical advice or instruction. Read More
© 2025 Healthy Hemp Oil. | Design by ahsan